A donut-shaped protein breaks apart to start bacterial cell division
- Date:
- March 14, 2026
- Source:
- Universitat Autonoma de Barcelona
- Summary:
- Researchers have revealed how bacteria precisely control the genes that trigger cell division. The study shows that the MraZ protein, which normally forms a donut-shaped structure, must bend and partially break apart to bind key DNA sequences that activate division genes. Using cryo-electron microscopy, scientists captured this interaction in remarkable detail. The mechanism appears to be widespread across bacteria, offering a new window into how microbes regulate growth.
- Share:
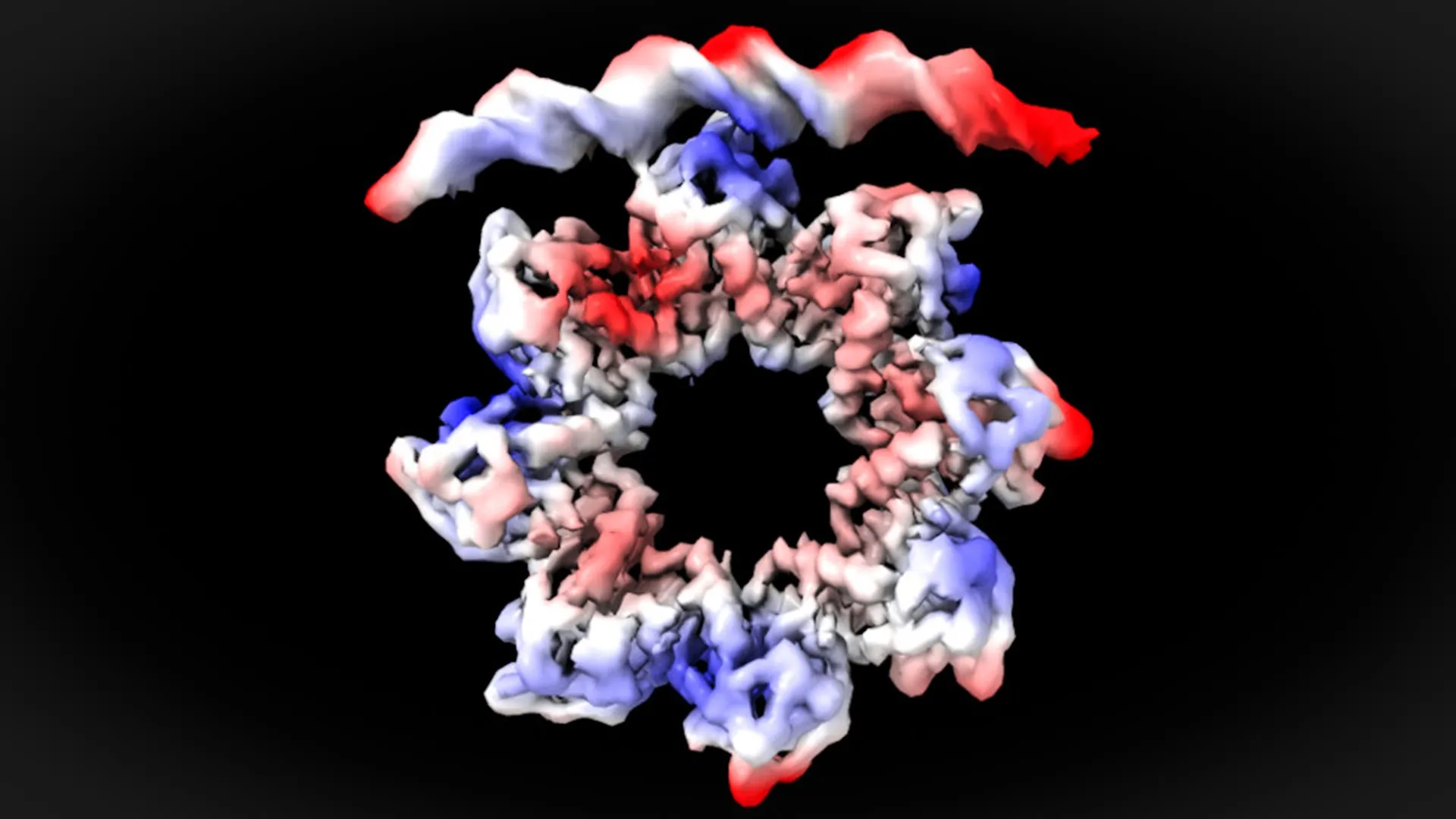
A research team led by Universitat Autònoma de Barcelona (UAB) scientist David Reverter has identified the molecular mechanism that regulates bacterial cell division. The discovery reveals how the MraZ protein binds to the dcw gene cluster to control this process. The findings were published in Nature Communications.
Cell division is essential for all living organisms and depends on the coordinated activity of many proteins and regulatory components. In most bacteria, the instructions for this process are organized within a group of genes known as the dcw operon. This cluster contains the genetic information needed to produce proteins responsible for both cell division and the construction of the bacterial cell wall.
How the dcw Operon Activates Cell Division Genes
The genes in this cluster are turned on by proteins known as transcription factors. These proteins attach to a specific section of DNA called the promoter, which marks where transcription begins. This starting point appears just before the first codon (the basic unit of gene information) that signals the beginning of the protein sequence.
One of these transcription factors is MraZ, which is also the first gene within the dcw operon in all bacteria. When MraZ activates the operon, the genes within the cluster produce the proteins required for bacteria to divide. In this way, MraZ acts as the key regulator controlling the activity of the operon that governs cell division in most bacterial species.
Imaging the Molecular Mechanism of Bacterial Division
The UAB research group, led by David Reverter, full professor in the Department of Biochemistry and Molecular Biology and researcher at the Institute of Biotechnology and Biomedicine of the UAB (IBB-UAB), uncovered the detailed mechanism behind this regulation. The team used advanced structural biology methods including X-ray crystallography and cryo-electron microscopy.
These techniques allowed the scientists to determine how the MraZ transcription factor attaches to the promoter of the dcw operon in the bacterium Mycoplasma genitalium. This microorganism is frequently used in laboratory studies because it possesses an extremely small genome.
Atomic-Level View of the MraZ Protein Binding DNA
The promoter region of the dcw operon contains four repeated segments, or "boxes," each composed of six nucleotides. These repeated DNA sequences play a key role in regulating transcription.
By examining the system with cryo-electron microscopy, the researchers were able to observe the interaction between the MraZ protein and the DNA bases of these four repeated boxes at nearly atomic resolution. Their observations showed that MraZ must undergo a structural change in order to bind successfully to the operon.
"This is a surprising observation. The MraZ protein is an octamer formed by eight identical subunits joined in the shape of a donut, but with a curvature that would never allow the union with the four 'boxes' of the promoter. However, to regulate cell division we see how the donut breaks and deforms in such a way that four of the subunits can join the four boxes of the promoter," David Reverter explains.
A Major Advance in Understanding Bacterial Cell Division
Directly visualizing how MraZ interacts with the promoter DNA that initiates cell division represents a significant breakthrough. Until now, researchers studying this system relied largely on biochemical experiments and computer modeling to infer how the mechanism worked.
According to Reverter, the regulatory system identified in this study is likely widespread among bacteria. "is universal to most bacteria, since all MraZ proteins are very similar, have the same octamer structure, and the DNA sequences of the promoters of the operons that regulate cell division are also similar," Reverter concludes.
International Collaboration Behind the Study
The research was led by David Reverter's team at the Institute of Biotechnology and Biomedicine and the Department of Biochemistry and Molecular Biology at UAB. The work was carried out in collaboration with the ALBA synchrotron and the cryo-electron microscopy service of the Institute of Genetics and Molecular and Cellular Biology of Strasbourg, France.
Story Source:
Materials provided by Universitat Autonoma de Barcelona. Note: Content may be edited for style and length.
Journal Reference:
- Lucía Sánchez-Alba, Nathalia Varejão, Alexandre Durand, Javier García-Pardo, Maria Carreras-Caballé, Virginia Amador, Jaume Pinyol, David Reverter. Structural basis for transcriptional regulation by the cell division regulator MraZ in Mycoplasma genitalium. Nature Communications, 2026; 17 (1) DOI: 10.1038/s41467-026-68809-2
Cite This Page: