This “rotten egg” brain gas could be the key to fighting Alzheimer’s disease
Researchers found that a protein responsible for producing trace amounts of hydrogen sulfide is essential for memory and brain health.
- Date:
- April 8, 2026
- Source:
- Johns Hopkins Medicine
- Summary:
- Scientists have uncovered a surprising new player in Alzheimer’s disease: a protein called CSE that helps produce tiny amounts of hydrogen sulfide gas in the brain. In experiments with genetically engineered mice, removing this protein led to memory loss, brain damage, and other hallmarks of Alzheimer’s, including weakened blood-brain barriers and reduced formation of new neurons. The findings suggest that this “rotten egg” gas, when carefully regulated, may actually protect brain cells and support memory.
- Share:
Researchers at Johns Hopkins Medicine report that a newly funded study by the National Institutes of Health is helping advance a potential new approach to Alzheimer's disease treatment. The focus is a protein in the brain that produces a small but important gas.
The protein, called Cystathionine γ-lyase, or CSE -- best known for generating hydrogen sulfide, the gas that smells like rotten eggs -- appears to play a key role in how memory forms. The findings come from experiments in genetically engineered mice, according to study leader Bindu Paul, M.S., Ph.D., associate professor of pharmacology, psychiatry and neuroscience at the Johns Hopkins University School of Medicine.
The research, published in Proceedings of the National Academy of Sciences, aims to better understand how this protein works and whether boosting its activity could help protect brain cells and slow neurodegenerative diseases such as Alzheimer's.
Hydrogen Sulfide May Protect Brain Cells
Earlier studies suggested that hydrogen sulfide can help protect neurons in mice. However, the gas is toxic in large amounts, which makes it unsafe to deliver directly to the brain. Scientists are instead trying to understand how to safely maintain the extremely small levels naturally present in neurons.
The new findings show that mice engineered to lack the CSE enzyme develop problems with memory and learning. These mice also show increased oxidative stress, DNA damage and weakened blood-brain barrier integrity -- all features commonly associated with Alzheimer's disease, says Paul, the study's corresponding author.
Building on Years of Research
The current work builds on earlier research led by Solomon Snyder, M.D., D.Sc., D.Phil., professor emeritus of neuroscience, pharmacology, and psychiatry. In 2014, his team reported that CSE supported brain health in mice with Huntington's disease. The researchers used mice lacking the CSE protein, first developed in 2008 when the protein was linked to blood vessel function and blood pressure regulation.
In 2021, the group found that CSE was not functioning properly in mice with Alzheimer's disease, and that very small injections of hydrogen sulfide helped protect brain function.
Those earlier studies focused on mice with additional genetic mutations tied to neurodegenerative diseases. The latest research isolates the role of CSE itself.
"This most recent work indicates that CSE alone is a major player in cognitive function and could provide a new avenue for treatment pathways in Alzheimer's disease," says co-corresponding author Snyder, who retired from the Johns Hopkins Medicine faculty in 2023.
Memory Loss Linked to CSE Deficiency
To better understand how CSE affects memory, scientists compared mice lacking the protein with normal mice using the same strain developed in 2008. They tested spatial memory (ability to remember directions and follow cues) using a setup called the Barnes maze.
In this test, mice learn to escape a bright light by finding a hidden shelter. At two months old, both normal mice and those lacking CSE performed similarly, locating the shelter within three minutes. By six months, however, the CSE-deficient mice struggled to find the escape route, while normal mice continued to succeed.
"The decline in spatial memory indicates a progressive onset of neurodegenerative disease that we can attribute to CSE loss," says first author Suwarna Chakraborty, a researcher in Paul's lab.
Brain Changes Mirror Alzheimer's Disease
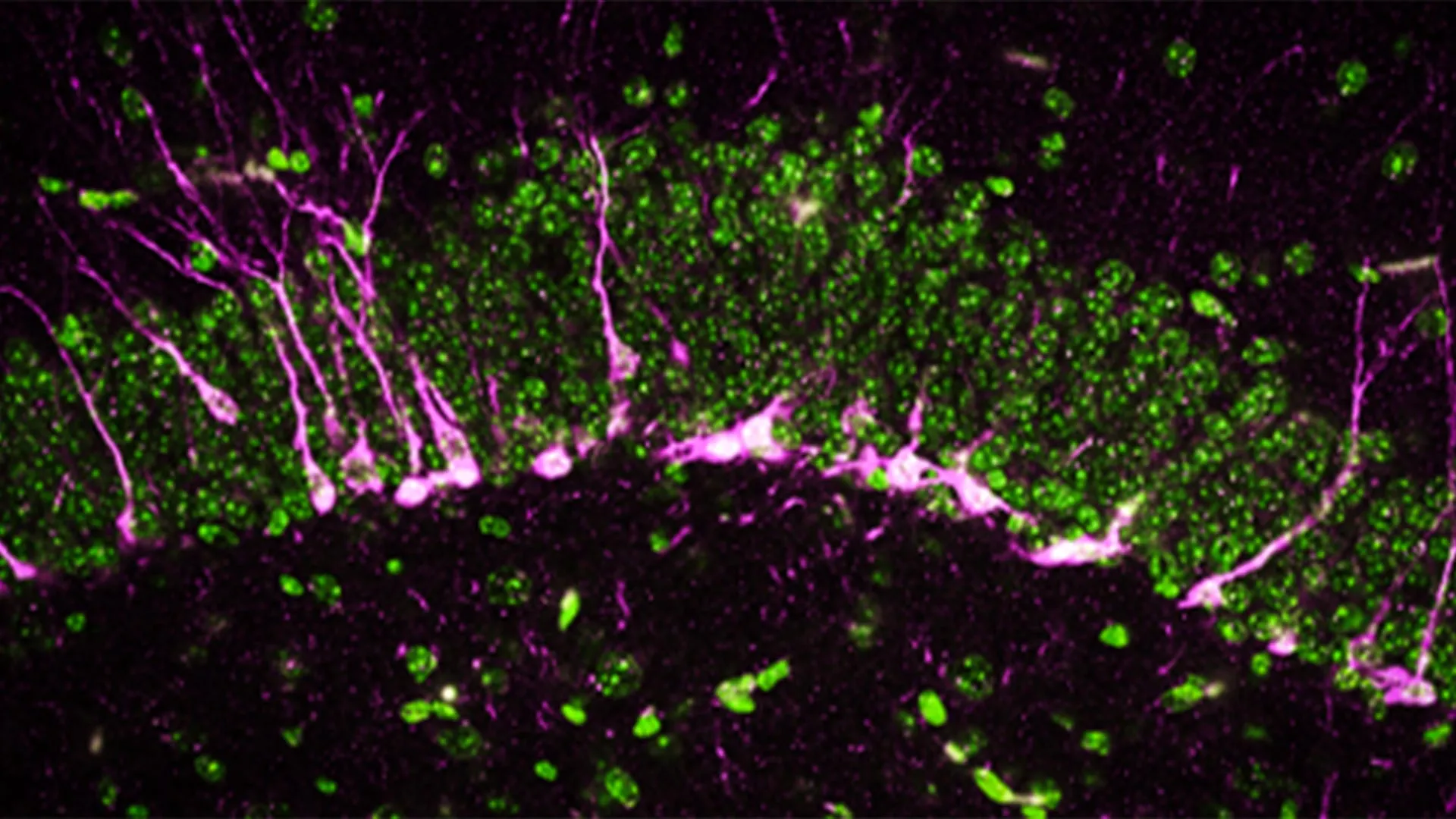
The researchers also examined how the absence of CSE affects the brain at a cellular level. The hippocampus, a region critical for learning and memory, relies on the formation of new neurons. Disruptions in this process are a known feature of neurodegenerative diseases.
Using biochemical and analytical methods, the team found that proteins involved in neurogenesis were reduced or missing in mice without CSE.
With high powered electron microscopes, the scientists observed structural damage in the brains of these mice. They found large breaks in blood vessels, indicating harm to the blood-brain barrier, another hallmark of Alzheimer's disease. In addition, newly formed neurons had difficulty reaching the hippocampus, where they normally contribute to memory formation.
"The mice lacking CSE were compromised at multiple levels, which correlated with symptoms that we see in Alzheimer's disease," says co-first author Sunil Jamuna Tripathi, a researcher in Paul's lab.
Toward New Alzheimer's Treatments
Alzheimer's disease affects more than 6 million people in the United States, according to the U.S. Centers for Disease Control and Prevention, and the number continues to grow. Currently, no treatments have been consistently shown to stop or slow the disease.
The researchers say that targeting CSE and its production of hydrogen sulfide could offer a new path for developing therapies aimed at protecting brain function and slowing disease progression.
Funding and Research Contributors
Funding support for this research was provided by the National Institutes of Health (1R01AG071512, P50 DA044123,1R21AG073684, O1AGs066707, U01 AG073323, AG077396, NS101967, NS133688, P01CA236778), the Department of Defense (HT94252310443), the American Heart Association, AHA-Allen Initiative in Brain Health and Cognitive Impairment, the Solve ME/CFS Initiative, the Catalyst Award from Johns Hopkins University, the Valour Foundation, the Wick Foundation, Department of Veterans Affairs Merit Award (I01BX005976), the Louis Stokes Cleveland Department of Medical Affairs Veterans Center, the Mary Alice Smith Funds for Neuropsychiatry Research, the Lincoln Neurotherapeutics Research Fund, the Gordon and Evie Safran Neuropsychiatry Fund; and the Leonard Krieger Fund of the Cleveland Foundation.
In addition to Paul, Snyder, Chakraborty and Tripathi, contributors included Richa Tyagi and Benjamin Orsburn from Johns Hopkins; Edwin Vázquez-Rosa, Kalyani Chaubey, Hisashi Fujioka, Emiko Miller and Andrew Pieper of Case Western University; Thibaut Vignane and Milos Filipovic from Leibniz Institute for Analytical Sciences, Germany; Sudarshana Sharma from Hollings Cancer Center; Bobby Thomas from Darby Children's Research Institute and the Medical University of South Carolina, and Zachary Weil and Randy Nelson from West Virginia University School of Medicine.
Story Source:
Materials provided by Johns Hopkins Medicine. Note: Content may be edited for style and length.
Journal Reference:
- Suwarna Chakraborty, Sunil Jamuna Tripathi, Edwin Vázquez-Rosa, Kalyani Chaubey, Hisashi Fujioka, Emiko Miller, Richa Tyagi, Thibaut Vignane, Sudarshana M. Sharma, Bobby Thomas, Zachary M. Weil, Randy J. Nelson, Milos R. Filipovic, Benjamin C. Orsburn, Solomon H. Snyder, Andrew A. Pieper, Bindu D. Paul. Cystathionine γ-lyase is a major regulator of cognitive function through neurotrophin signaling and neurogenesis. Proceedings of the National Academy of Sciences, 2025; 122 (52) DOI: 10.1073/pnas.2528478122
Cite This Page: