Scientists stunned as bacteria rewire DNA machinery to shape cells
Ancient bacteria rewired a DNA system into a shape-controlling “skeleton”—a striking example of evolution’s creativity.
- Date:
- April 20, 2026
- Source:
- Institute of Science and Technology Austria
- Summary:
- Cyanobacteria—ancient microbes that oxygenated Earth and made complex life possible—are still revealing surprises billions of years later. Scientists have now discovered that a molecular system once used to separate DNA has been repurposed into something entirely different: a structure that shapes the cell itself.
- Share:
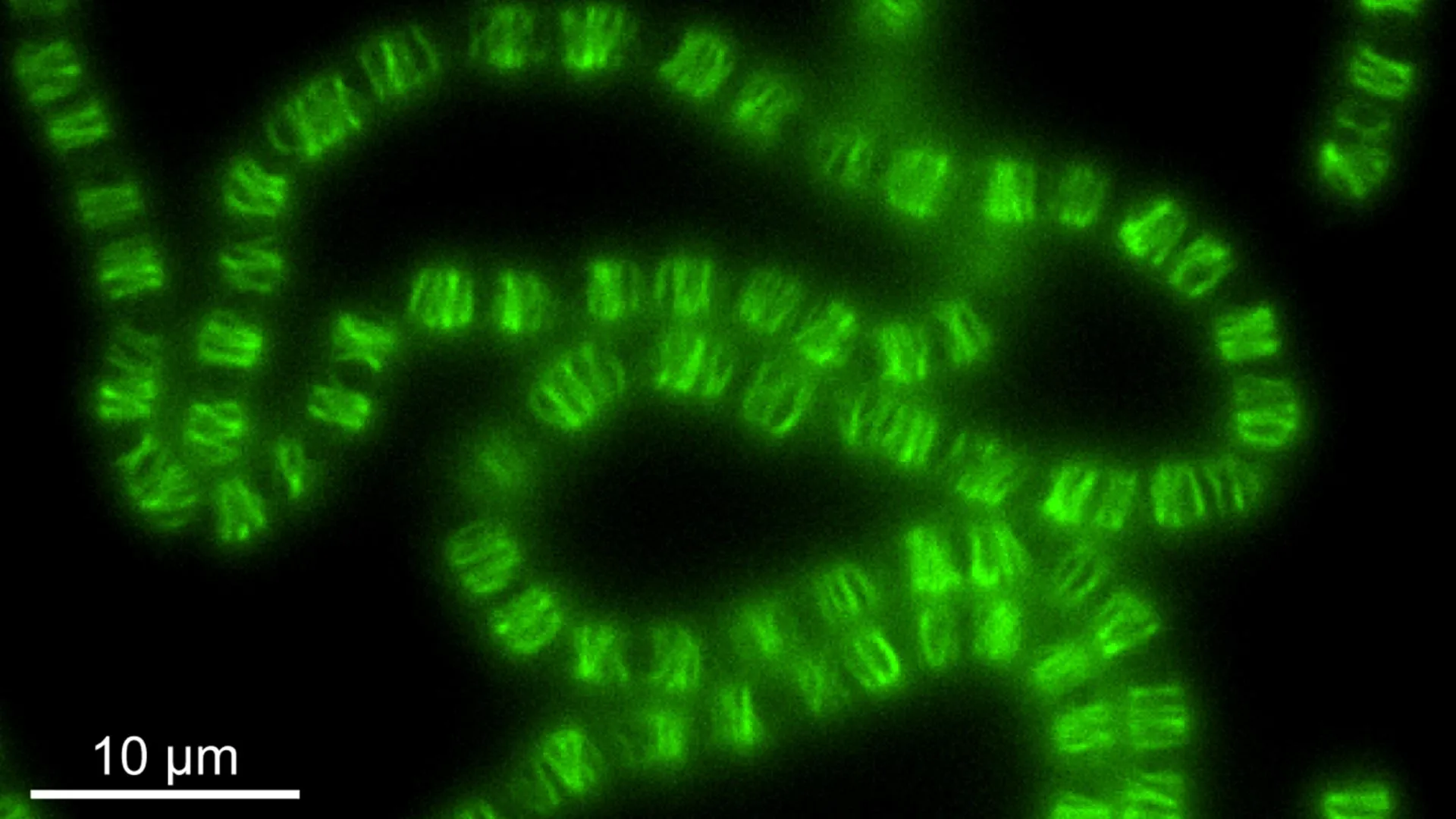
Photosynthetic bacteria played a major role in shaping Earth as we know it. Among them, cyanobacteria stand out for producing the oxygen that filled our atmosphere and allowed complex life to emerge. Now, scientists at the Institute of Science and Technology Austria (ISTA) have uncovered a surprising twist in how these organisms work. A biological system once believed to separate DNA has instead evolved to control the shape of cyanobacterial cells. The findings, published in Science, offer new insight into how protein systems change over time and how multicellular life developed in these ecologically important bacteria.
"Cyanobacteria are essentially pioneers of oxygenic photosynthesis," says Benjamin Springstein, a postdoc in the Loose group at the Institute of Science and Technology Austria (ISTA).
"They are responsible for the Great Oxygenation Event about 2.5 billion years ago, when oxygen accumulated in the atmosphere and made aerobic life possible. Without them, it's safe to say that none of us would be here today."
Even today, cyanobacteria remain essential to life on Earth. They contribute heavily to global biomass and play central roles in carbon and nitrogen cycles. These organisms are highly adaptable, living in extreme conditions ranging from hot springs to the Arctic, as well as on surfaces like roofs and walls in cities. One species in particular, Anabaena sp. PCC 7120 (or simply Anabaena), has been studied for over three decades and serves as a model for understanding multicellular cyanobacteria.
Evolution Turns DNA System Into Cell-Shaping Structure
Springstein worked with Professor Martin Loose's group alongside collaborators from ISTA, the Institut Pasteur de Montevideo (Uruguay), Kiel University (Germany), and the University of Zürich (Switzerland). Together, they found that Anabaena, and likely other multicellular cyanobacteria, have undergone a major evolutionary shift. An ancient system once used to separate DNA during cell division has been repurposed into a cytoskeleton-like structure that helps determine cell shape.
DNA in Bacteria Explained
Like all bacteria, Anabaena reproduce by dividing into new cells. This process depends on accurately copying and distributing DNA so that each new cell receives the genetic information it needs to survive. DNA is tightly packed into chromosomes, similar to thread wound around a spool, and is often present in multiple copies that must be reliably passed on during division.
Bacterial DNA comes in two main forms. Chromosomes contain essential genes required for survival, while plasmids carry additional genes that are often not essential. Plasmids can move between bacteria, allowing traits to spread quickly and enabling rapid adaptation.
A DNA Segregation System With a New Role
Springstein has studied Anabaena since 2014, exploring its biology and evolution. During the COVID-19 pandemic, when lab work paused, he spent time reviewing scientific literature and noticed something unexpected.
"I made a serendipitous observation," he recalls.
He found that Anabaena and some related cyanobacteria contain a system known as ParMR encoded within their chromosomes. Traditionally, this system is linked to plasmid segregation and had only been found on plasmids, which are mobile genetic elements. This unusual placement led him to suspect that the system might have adapted to separate chromosomes instead.
After joining ISTA as an IST-Bridge Fellow, Springstein set out to test this idea. His experiments revealed something very different. One component, ParR, no longer binds to DNA. Instead, it attaches to lipid membranes, especially the inner membrane of the cell. Meanwhile, ParM does not form structures in the cytoplasm to move DNA. Instead, it creates filament networks just beneath the inner membrane, forming a layer of protein polymers that resembles a cell cortex.
Rather than acting like a typical DNA segregation system that forms spindle-like structures in the cell interior, this system operates at the membrane level and appears to organize cell structure.
Filaments That Behave Like a Cellular Skeleton
To better understand how this system works, researchers recreated it outside living cells using purified components. In these in vitro reconstitution experiments, they observed that the filaments display dynamic instability. They grow and then rapidly collapse, a behavior similar to microtubules in more complex cells.
To investigate further, the team collaborated with ISTA Professor Florian Schur and his PhD student Manjunath Javoor. Using cryo-electron microscopy, which allows scientists to see molecular structures in great detail, they examined how these filaments are built. They discovered that, unlike the polar filaments formed by similar systems in other bacteria, the filaments in Anabaena are bipolar, meaning they can grow and shrink from both ends.
Loss of the System Alters Cell Shape
The true function of this system became clear when it was removed from living cells.
"Cells lacking the system lost their normal rectangular-like cell shape and instead became round and swollen," Springstein explains.
These kinds of changes are typically seen when genes responsible for maintaining cell shape are disrupted in other bacteria. This strongly suggests that the system's main role is to control cell structure rather than manage DNA distribution.
Given its new function and location in the cell, the researchers renamed the system "CorMR."
How Evolution Repurposed an Ancient System
Multicellular cyanobacteria evolved gradually from single-celled ancestors, gaining complexity over time. Bioinformatic analysis by collaborator Daniela Megrian from the Institut Pasteur in Montevideo, Uruguay, helped clarify how the CorMR system came to be.
The transformation did not happen all at once. Instead, it likely occurred through a sequence of changes. First, the system shifted from a plasmid to the chromosome. Next, its components changed in size and structure. Then, it developed the ability to bind to cell membranes. Finally, it came under the control of an additional protein system.
Together, these steps converted an ancient DNA segregation mechanism into a system that shapes the cell itself, offering a striking example of how evolution can give old biological tools entirely new purposes.
Story Source:
Materials provided by Institute of Science and Technology Austria. Note: Content may be edited for style and length.
Journal Reference:
- Benjamin L. Springstein, Manjunath G. Javoor, Daniela Megrian, Roman Hajdu, Dustin M. Hanke, Bettina Zens, Gregor L. Weiss, Florian K. M. Schur, Martin Loose. Repurposing of a DNA segregation machinery into a cytoskeletal system controlling cell shape. Science, 2026; 392 (6795) DOI: 10.1126/science.aea6343
Cite This Page: